Article updated on:
April 15, 2026
DVC Stem employs a dedicated team of medical professionals, tasked with verifying the accuracy of health claims and summaries of medical research. Each member's expertise is aligned with the subject matter of the article to ensure precision and relevance.

Fact-Checked
We evaluate medical studies published in reputable scientific journals to form our opinions on a product or health matter, ensuring the utmost scientific precision.

Medically Cited
Mesenchymal Stem Cells Cross the Blood Brain Barrier (BBB)
Modern studies suggest that Mesenchymal stem cells (MSCs) when administered intravenously, can cross the blood-brain barrier (BBB) leading to various regenerative effects.
Learn more about our Stem Cell Treatment
Our treatment protocol is aimed at revolutionizing treatments for degenerative conditions.
50,000+ subscribers
Join our newsletter to learn more about stem cell therapy and the science behind it.
Yes — research has demonstrated that mesenchymal stem cells (MSCs) can cross the blood-brain barrier (BBB), making them particularly relevant for treating neurological conditions like MS, Alzheimer's, Parkinson's, and traumatic brain injury. MSCs use several mechanisms to navigate this protective barrier. Here's how it works and why it matters.
Mesenchymal Stem Cells Crossing the Blood-Brain Barrier
Mesenchymal stem cells (MSCs), when administered intravenously, have shown the ability to cross the blood-brain barrier (BBB). These cells have inherent properties that enable them to migrate toward inflammatory areas.
MSCs release various secretomes that promote nerve regeneration and provide pain relief at these sites. Research, including a study by Kim et al., has confirmed that MSCs administered intravenously can traverse the BBB, as evidenced in a mouse model for Alzheimer's Disease, demonstrating their potential for regenerative effects in the brain Study on MSCs and BBB.
Key Attributes of MSCs
- Inflammation Homing: MSCs naturally migrate to sites of inflammation.
- Secretomes Release: They secrete substances aiding in nerve regeneration and pain relief.
- BBB Crossing Capability: Intravenous administration of MSCs allows them to cross the BBB.
Understanding the Blood-Brain Barrier
The blood-brain barrier (BBB) serves as a critical protective layer around the brain and spinal cord. It regulates the entry and exit of substances to and from the brain. Composed of brain endothelial cells, which line the brain's blood vessels, these cells form tight connections known as junctional adhesion molecules with adjacent cells, creating a robust barrier.
Components and Function of the BBB
- Brain Endothelial Cells: Form the primary structure of the BBB.
- Tight Junctions: These junctions between cells ensure a strong barrier.
- Selective Permeability: Controls what substances can enter or leave the brain.
Blood-Brain Barrier's Impact on Stem Cell Therapy
The blood-brain barrier (BBB) is a critical structure in the brain, consisting of a complex network of blood vessels and cells. It acts as a protective shield, controlling the passage of substances between the bloodstream and the central nervous system (CNS).
This barrier is essential for maintaining brain homeostasis and plays a vital role in the efficacy of stem cell therapies for neurological conditions NIH PMC.
Factors Influencing Stem Cell Delivery Across the BBB
- Barrier Function: The BBB can block certain stem cells from entering the CNS, affecting their therapeutic potential. A study showed that human dermis-derived mesenchymal stem cells injected early after transient global cerebral ischemia did not cross a damaged BBB PubMed.
- Timing of Administration: The success of stem cell therapy is influenced by when the cells are administered in relation to BBB damage. Research in a Temporal Lobe Epilepsy model indicated that the strategic timing of administering DPSCs or BM-MSCs following BBB damage due to status epilepticus enhanced CNS homing and reduced neurodegeneration and other complications PubMed.
- Role of Extracellular Vesicles: Mesenchymal stem cell-derived extracellular vesicles, like exosomes, can cross the BBB. These vesicles hold therapeutic promise and can be delivered via intravenous or intranasal routes, avoiding invasive neurosurgical procedures NIH PMC.
The BBB significantly influences the delivery and effectiveness of stem cells in treating neurological disorders. A thorough understanding of the BBB's functioning and strategic timing of stem cell administration is key to optimizing their therapeutic potential.
Further Study Data
Mesenchymal stem cells (MSCs) are increasingly recognized for their ability to traverse the blood-brain barrier (BBB), presenting new avenues for treating neurological disorders.
Notably, in a Temporal Lobe Epilepsy (TLE) animal model, dental pulp stem cells (DPSCs) and bone marrow mesenchymal stem cells (BM-MSCs) were effective in crossing the BBB when administered systemically following status epilepticus-induced BBB damage.
This facilitated CNS homing and reduced neurodegeneration, neuroinflammation, and neuropsychiatric issues PubMed.
Interactions
- MSCs in Neurological Treatment: Their potential in addressing various neurological conditions is significant.
- Effectiveness in TLE Model: Timed administration of DPSCs and BM-MSCs showed promising results in mitigating neurological complications in TLE.
- Intranasal and Intravenous Delivery: Extracellular vesicles (EVs) from MSCs, including exosomes, can cross the BBB. They offer therapeutic potential in clinical settings, with delivery possible via intravenous or intranasal routes, avoiding neurosurgery NIH PMC.
Mesenchymal stem cells have demonstrated the capability to cross the blood-brain barrier, showing potential in alleviating neurological conditions in various animal models. However, the success of MSCs in BBB penetration may vary depending on the specific condition and timing of administration.
Brain Capillary Endothelial Cells and Nutrient Regulation
Brain capillary endothelial cells are a key part of the BBB. They help maintain brain metabolism and support neuronal function by selectively allowing essential nutrients, such as glucose and amino acids, to pass through while keeping out harmful substances. They also regulate cerebral blood flow, which is crucial for providing oxygen and nutrients to the brain.
Vascular Endothelial Growth Factor and BBB Formation
A critical factor in the formation and function of the BBB is the vascular endothelial growth factor (VEGF). This protein helps create and maintain the brain capillary endothelium, essential for the BBB's barrier function. The BBB also prevents most immune cells, like white blood cells and T cells, from entering the brain, which helps to protect the neural tissue from inflammation and damage.
Circumventricular Organs and Weak Points in the BBB
However, there are some areas of the brain, known as circumventricular organs, where the BBB is weaker, allowing certain substances to enter more easily. For example, lipid-soluble molecules and fat-soluble molecules can pass through the BBB, while other substances, like essential amino acids, use unique transport systems to cross.
The Cerebrospinal Fluid Barrier and Brain Support
The cerebrospinal fluid (CSF) is another critical component of the BBB. It surrounds the brain and spinal cord, providing structural support and helping to remove waste products. The cerebrospinal fluid barrier is formed by epithelial cells that line the brain's ventricles, creating a separation barrier between the CSF and the circulating blood.
Disruptions in BBB Function and Effects on Brain Health
The BBB's function can be disrupted in certain situations, such as brain tumors or ischemic stroke, leading to increased BBB permeability. This allows substances from the blood to enter the brain, potentially causing harm to brain cells and affecting brain function. Maintaining the integrity of the BBB is crucial for overall brain health and the proper functioning of the nervous system.
Stem Cells and Cellular Therapy for Neurodegenerative Diseases
Stem cells, including mesenchymal, neural, and embryonic stem cells, are reported to bypass the blood-brain barrier (BBB) and successfully home to their target in the brain, making them a promising approach in cellular therapy for neurodegenerative diseases.
Crossing the Blood-Brain Barrier
The blood-brain barrier acts like a security checkpoint, keeping harmful substances out of the brain. However, stem cells can cross this barrier. When an injury or disease occurs in the brain, the BBB becomes weaker, allowing stem cells to slip through and reach the affected area.
Although the BBB is a selective barrier, stem cells can migrate across endothelial cells through either the paracellular or transcellular pathway and subsequently preferentially home back to the site of inflammation or injury in the brain to exert their therapeutic effects. In CNS injuries such as traumatic brain injury, stroke, brain tumor, or aging, lower tight junction integrity and formation of paracellular gaps allow cell migration via the paracellular route. MSCs have also been reported to abolish and split tight junctions between endothelial cells.
Systemic Homing Process of MSCs
When stem cells are introduced into a patient's body, they enter the bloodstream and begin a multi-step process to reach their target. First, they slow down and attach to the inner walls of blood vessels. Then, they follow chemical signals from particular proteins that guide them to the correct location.
The systemic homing of MSCs is believed to result from their interaction with endothelial cells in a leukocyte-like, multistep cascade. MSCs first enter the bloodstream when they are therapeutically administered. The homing process begins with MSCs decelerating and coming into contact with the endothelial wall by tethering to the selectins expressed by endothelial cells and starting to roll along the vasculature wall. Next, integrin receptors like VLA-4 are activated in response to G-protein-coupled chemokine receptors such as stromal cell-derived factor-1 (SDF-1), which binds ligands expressed by MSCs such as CXCR4 or CXCR7. Following integrin activation, MSCs arrest the endothelial membrane as integrins like VLA-4 expressed by MSC bind with VCAM-1 on endothelial cells. The onetime activation increases the affinity of integrins essential for cell arrest; hence, the VLA-4/VCAM-1 interaction allows MSCs to adhere to the endothelial cells firmly.
Transmigration and Migration to Injury Site
Once stem cells are near the damaged area of the brain, they need to exit the blood vessels and move into the brain tissue. They do this by squeezing through the walls of the blood vessels, a process called transmigration. After they've left the blood vessels, the stem cells follow more chemical signals to find the exact spot where they're needed. Once they're in the right place, they can start working to repair the damage.
Next, MSCs travel through the endothelial cell layer and basement membrane in transmigration, facilitated by the secretion of matrix metalloproteinases (MMPs) which degrade the endothelial basement membrane. Alternatively, abolishing and splitting the tight junctions between endothelial cells by MSC could facilitate their transmigrations. MSCs have also been reported to penetrate the endothelial via plasmic podia. Finally, MSCs migrate to the injury site, guided by various signals the damaged tissue releases, such as growth factors and chemokines.
Stem cells cross the blood-brain barrier in an animal stroke model
Mesenchymal stem cells represent a great potential to reverse neuronal damage associated with CNS diseases such as Multiple Sclerosis, Parkinson's disease (PD), and Alzheimer's disease (AD). (4)
A study conducted by Yilmaz et al. found that intravenously (IV) injected mesenchymal stem cells (MSCs) can travel through the blood-brain barrier to the cerebral artery occlusion (t-MCAO) model for stroke. (5) The brain tropism (movement into the brain) of MSCs was confirmed by whole-body imaging of radiolabeled (visible radioactive compound) MSCs given to rats.
During the first two hours after stroke, MSCs transiently pass through the lungs and continue to migrate over time within the region of brain ischemia, crossing the blood-brain barrier. The study concluded through whole-body imaging that mesenchymal stem cells (MSCs) when administered intravenously, could populate the CNS after passing through the BBB. (5)
What makes mesenchymal stem cells (MSCs) so special?
Mesenchymal stem cells (MSCs) are a beautiful therapeutic agent due to their ease of isolation, established safety, and potential to target multiple pathways involved in neuronal regeneration.
MSCs are widely used in treating various diseases due to their self-renewable, differentiation, anti-inflammatory, and immunomodulatory properties. In-vitro (performed in a laboratory setting) and in-vivo (taking place in a living organism) studies have supported the understanding of the mechanisms, safety, and efficacy of MSC therapy in clinical applications. (6)
Do mesenchymal stem cells get trapped in the lungs?
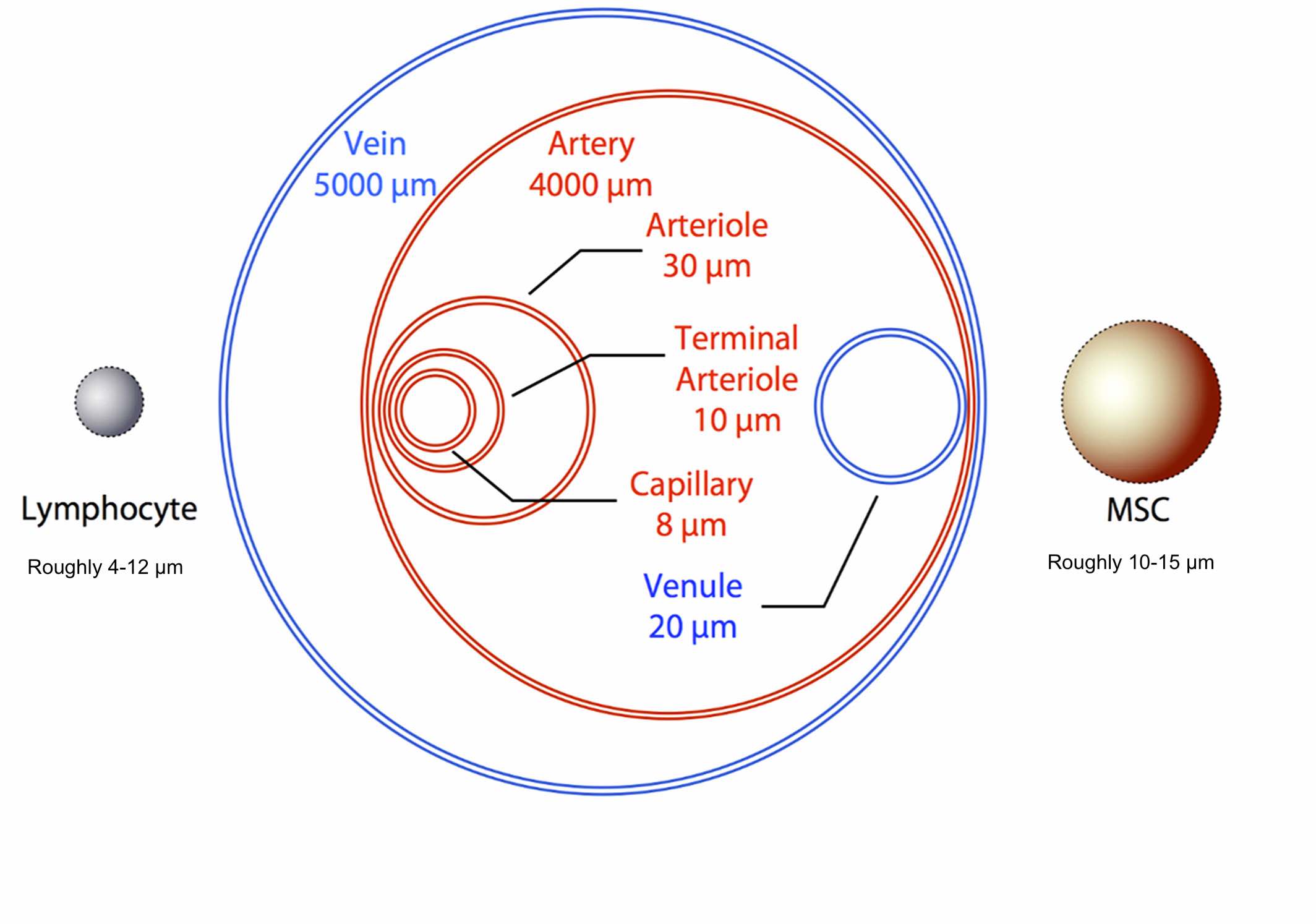
Studies suggest that pulmonary trapping of stem cells following intravenous administration is only a transient phenomenon, meaning the cells eventually pass through reaching other body areas (11). Physical size plays a vital role in the migratory capabilities of any cell throughout the body. (see figure below).
The size of cells can result in entrapment in certain areas resulting in a loss of migratory abilities. Mesenchymal stem cells (MSCs) can range in size depending on their source (bone marrow, adipose tissue, cord blood, or umbilical cord tissue). Importantly, endogenous MSCs are smaller (∼10 μm), enabling efficient trafficking via systemic circulation (10).
According to a 2009 study conducted by Major et al. the average diameter of a single cord tissue-derived mesenchymal stem cell is roughly 11 μm (similar in size to a white blood cell) (9). Mesenchymal stem cells also undergo cellular deformability, which can facilitate the passage of larger cells through smaller vessels (10). This data suggests that MSCs can bypass the pulmonary' first-pass effect' (becoming trapped in the lungs) leading to efficient circulation throughout the entire body, including the CNS.
Homing mechanisms of mesenchymal stem cells (MSCs)
One of the critical benefits of mesenchymal stem cells is their ability to target specific areas of concern due to their intrinsic homing capabilities. Mesenchymal stem cell homing, when administered systemically, can be defined as exiting circulation and migrating to the injury site. (7)
Studies suggest that MSCs may possess leukocyte-like, active homing mechanisms that enable them to interact with and migrate across the BBB under injury or inflammation. (1)
Stem cells administered intravenously can cross the blood-brain barrier
Neurological diseases are generally difficult to treat, partly due to the challenge of getting drugs across the blood-brain barrier (BBB). However, when administered via IV, mesenchymal stem cells can cross the blood-brain barrier (BBB). Studies have shown that mesenchymal stem cells rapidly migrate to damaged brain regions. This has been confirmed through magnetic resonance-based tracking of the transplanted cells. (1).
The opening into the brain (BBB) is believed to allow for the efficient crossing of MSCs and their secretome to the desired site in the brain, which results in various regenerative effects such as nerve regeneration, reduction in inflammation, and reduction in pain.
We have found multiple studies focusing on neurological conditions such as Multiple Sclerosis, Parkinson's, Stroke, and ALS, which have observed positive outcomes via intravenous injection of mesenchymal stem cells. (8) So, it can be determined that mesenchymal stem cells can cross the blood-brain barrier (BBB) in multiple different disease models. (4, 3, 5)
The Importance of the Blood-Brain Barrier in Brain Health
The blood-brain barrier (BBB) is a crucial central nervous system component, playing a vital role in protecting the brain and maintaining brain health. It is a complex vascular organ composed of brain capillaries, glial cells, integral membrane proteins, and cellular junctions, which control the brain uptake of nutrients and exclude harmful substances.
The BBB ensures that only small molecules and essential substances like fatty acids can penetrate the brain, while harmful elements, such as tumor necrosis factor and reactive oxygen species, are kept out. The barrier is formed by a continuous layer of cells lining the luminal surface of cerebral vessels and is supported by adjacent cells, such as glial cells.
Proper BBB function is essential for brain development, neuronal excitability, and the overall health of the nervous tissue. Dysfunction of the BBB can lead to various issues, including inflammation and damage to the brain. Therefore, understanding the cell physiology of the BBB and how it interacts with the vascular system is crucial for the development of therapies to treat and prevent brain disorders.
Research into the BBB has provided valuable insights into brain health, and continued study in this area, such as investigating animal brains and examining the role of the pineal gland in cardiovascular regulation, will further our understanding of this complex and essential barrier. By maintaining the integrity of the blood-brain barrier, we can help ensure the proper functioning of the brain and promote overall brain health.

Stem Cells and Their Ability to Cross the Blood-Brain Barrier
In conclusion, stem cells are promising for treating various neurological disorders, as they can cross the blood-brain barrier (BBB) and reach specific brain regions. While the BBB typically restricts the passage of most substances, stem cells can achieve brain penetration by using different mechanisms.
One such mechanism involves the interstitial fluid, which surrounds the cells and can act as a medium for stem cells to bypass the barrier. Additionally, stem cells can interact with the luminal membrane of the BBB and the cells that line the blood vessels, enabling them to enter the brain tissue.
In some cases, stem cells can take advantage of BBB dysfunction, which can occur in certain neurological disorders or injuries. This allows the stem cells to more easily infiltrate the affected brain regions and exert their therapeutic effects.
Our understanding of how stem cells cross the BBB has been significantly advanced through research, including studies published in prestigious scientific journals like Cold Spring Harbor Perspectives. As our knowledge grows, we can develop more effective stem cell therapies to treat various neurological conditions, ultimately improving patients' brain health and quality of life.
References:
(1) Liu, L., Eckert, M. A., Riazifar, H., Kang, D.-K., Agalliu, D., & Zhao, W. (2013). From blood to the brain: can systemically transplanted mesenchymal stem cells cross the blood-brain barrier? Stem cells international. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3753739/.
(2) Conaty P;Sherman LS;Naaldijk Y;Ulrich H;Stolzing A;Rameshwar P; (n.d.). Methods of Mesenchymal Stem Cell Homing to the Blood-Brain Barrier. Methods in molecular biology (Clifton, N.J.). https://pubmed.ncbi.nlm.nih.gov/30196403/.
(3) Kim, S., Chang, K.-A., Kim, J. a, Park, H.-G., Ra, J. C., Kim, H.-S., & Suh, Y.-H. (2012). The preventive and therapeutic effects of intravenous human adipose-derived stem cells in Alzheimer's disease mice. PloS one. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3458942/.
(4) Trounson, A. (2009, June 11). New perspectives in human stem cell therapeutic research. BMC medicine. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2702289/.
(5) Yilmaz, G., Vital, S., Yilmaz, C. E., Stokes, K. Y., Alexander, J. S., & Granger, D. N. (2011, March). Selectin-mediated recruitment of bone marrow stromal cells in the postischemic cerebral microvasculature. Stroke. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3042505/.
(6) Cona, L. A. (n.d.). Types of Mesenchymal Stem Cells (MSCs) and their Mechanisms of Action. RSS. https://www.dvcstem.com/post/mscs.
(7) Torres Crigna, A., Daniele, C., Gamez, C., Medina Balbuena, S., Pastene, D. O., Nardozi, D., … Bieback, K. (2018, June 15). Stem/Stromal Cells for Treatment of Kidney Injuries With Focus on Preclinical Models. Frontiers in medicine. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6013716/.
(8) Petrou, P., Kassis, I., Levin, N., Paul, F., Backner, Y., Benoliel, T., . . . Karussis, D. (2020, November 30). Beneficial effects of autologous mesenchymal stem cell transplantation in active progressive multiple sclerosis. Retrieved February 23, 2021, from https://academic.oup.com/brain/article/143/12/3574/6012789?login=true
(9) Majore, I., Moretti, P., Hass, R., & Kasper, C. (2009, March 20). Identification of subpopulations in mesenchymal stem cell-like cultures from human umbilical cord. Cell communication and signaling : CCS. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2676292/.
(10) Krueger, T. E. G., Thorek, D. L. J., Denmeade, S. R., Isaacs, J. T., & Brennen, W. N. (2018, August 1). Concise Review: Mesenchymal Stem Cell‐Based Drug Delivery: The Good, the Bad, the Ugly, and the Promise. Stem Cells Translational Medicine (AlphaMed Press). https://stemcellsjournals.onlinelibrary.wiley.com/doi/full/10.1002/sctm.18-0024.
(11) Fischer, U. M., Harting, M. T., Jimenez, F., Monzon-Posadas, W. O., Xue, H., Savitz, S. I., Laine, G. A., & Cox, C. S. (2009, June). Pulmonary passage is a major obstacle for intravenous stem cell delivery: the pulmonary first-pass effect. Stem cells and development. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3190292/.









.jpg)
