Article updated on:
April 15, 2026
DVC Stem employs a dedicated team of medical professionals, tasked with verifying the accuracy of health claims and summaries of medical research. Each member's expertise is aligned with the subject matter of the article to ensure precision and relevance.

Fact-Checked
We evaluate medical studies published in reputable scientific journals to form our opinions on a product or health matter, ensuring the utmost scientific precision.

Medically Cited
What are Stem Cells?
Discover the fundamentals of stem cells and their transformative potential in modern medicine. Uncover the science behind these unique cells and their applications in various treatments.
Learn more about our Stem Cell Treatment
Our treatment protocol is aimed at revolutionizing treatments for degenerative conditions.
50,000+ subscribers
Join our newsletter to learn more about stem cell therapy and the science behind it.
Stem cells are the body's master cells — undifferentiated cells capable of developing into many different cell types and serving as an internal repair system. They can divide to replenish other cells, making them essential for growth, healing, and the emerging field of regenerative medicine. Here's what you need to know.
What are stem cells?
Stem cells are particular types of cells that can develop into many different types of cells in the body. They have specialized functions such as self-renewal through cell division and differentiation into specific types of specialized cells, such as red blood cells, insulin-producing cells, neurons, or other blood cells.
Stem cells are often called the body's " building blocks, " as they can potentially develop into any tissue or organ. They are found in various body parts, including the bone marrow, blood, and embryonic tissue. The pluripotency of stem cells allows them to construct any organism's cell.
According to a study conducted in 2019, Stem cells are undifferentiated cells found in the human body that have the potential to become any cell within an organism. They can also reproduce and replenish themselves, making them unique compared to other cell types. These cells can be found in both embryonic and adult stages. (1)
{{protocol2="/styling"}}
Stem cells are crucial for treating blood disorders, autoimmune and inflammatory conditions due to their ability to regulate the immune system and modulate inflammation. These unique cells in the human body can differentiate into different types including blood, bone, and muscle cells, and also have the capacity to repair damaged tissues. Currently, medical researchers are exploring stem cells' potential to differentiate into new tissues and treat a broad range of other diseases.
Importance of Stem Cells in Medicine and Research
Stem cells are crucial for their unique capabilities in medicine and scientific research. They offer potential in regenerative medicine, disease treatment, and understanding fundamental biological processes.
Key Benefits of Stem Cell Research
- Regenerative Potential: Stem cells can continuously self-renew and differentiate into various cell types, acting as a natural repair system for the body. This regenerative ability is essential for replacing lost or damaged cells throughout a person's life (NIH) (National Stem Cell Foundation).
- Disease Treatment: They are pivotal in treating numerous diseases and conditions like diabetes, heart disease, and neurodegenerative disorders, contributing significantly to the field of regenerative medicine (WebMD) (National Stem Cell Foundation).
- Drug Development: In the pharmaceutical industry, stem cells aid in drug development by allowing scientists to test new drugs and study disease progression in model systems (UNMC) (UNMC).
- Understanding Biology: Research on stem cells provides insights into cellular properties and development, enhancing our understanding of how organisms grow from a single cell and maintain healthy tissue (UNMC).
- Transplantation: Stem cells from bone marrow or umbilical cord blood are used in transplant therapies for blood cancers. Additionally, induced pluripotent stem cells (iPSCs) might be used for transplants without immune rejection risks (WebMD) (National Stem Cell Foundation).
Stem cells are considered to be of great importance in medical research due to their unique ability to differentiate into various types of cells and tissues, as well as their potential for self-renewal. This makes them useful in various therapeutic applications, including regenerative medicine, tissue engineering, and treating diseases such as cancer, Alzheimer's, MS, Crohn's, and heart disease. Studying stem cells can provide valuable insights into the development and progression of various diseases and may aid in developing new treatments and therapies. As such, stem cells have become a key area of focus in biomedical research, with many researchers and scientists working to unlock the full potential of these cells in the pursuit of improving human health.
What are the main types of stem cells
This section will discuss the different types of stem cells in the human body and their unique characteristics. T
hese types include embryonic stem cells, adult stem cells, and induced pluripotent stem cells. Understanding the different types of stem cells is crucial for understanding their potential uses in medical research and therapies.
Embryonic stem cells (Pluripotent stem cells)
These stem cells are derived from an early embryo stage of human development, typically from the blastocyst stage of an embryo.
They are pluripotent, meaning they have the potential to differentiate into any cell type of the body. Human pluripotent stem cells can make almost any cell in the body.
Characteristics
Embryonic stem cells are derived from the inner cell mass of a blastocyst, an early-stage embryo. They have the unique ability to differentiate into any cell type in the body and have the potential to self-renew.
This versatility makes them highly valuable for medical research, as they have the potential to be used to replace damaged or lost tissue in a variety of diseases and injuries.
Research and potential use
The embryonic stem cells used in research today come from unused embryos. These result from an in vitro fertilization procedure. Embryonic stem cells have been observed to have a high degree of pluripotency, meaning they can differentiate into many different cell types.
Using embryonic stem cells in research and therapeutic applications also poses certain risks. One primary concern is the potential for the development of tumors, as embryonic stem cells can divide and grow uncontrollably.
Additionally, embryonic stem cells may not be fully immunocompatible with the host, leading to rejection or an immune response. There are also ethical concerns and potential legal issues surrounding embryonic stem cells, as they are obtained from human embryos destroyed in the process.
Comparison of Embryonic Stem Cells and Adult Stem Cells
Embryonic and adult stem cells are both unspecialized cells capable of differentiating into various cell types in the body. Adult stem cells are generally preferred for clinical applications due to their accessibility, lower ethical concerns, and reduced health risks compared to embryonic stem cells.
Key Differences and Advantages of Adult Stem Cells
- Source of Cells: Adult stem cells are derived from tissues like bone marrow, umbilical cord, adipose tissue, and blood, making them more readily accessible and ethically acceptable.
- Risk of Tumors: Adult stem cells have a significantly lower risk of tumor formation as they are more mature and have limited division capabilities, unlike embryonic stem cells which can proliferate extensively and may lead to abnormal cell growth.
- Clinical Use: Due to their specific differentiation potential and safety profile, adult stem cells are more suitable for therapeutic use, offering targeted treatment options with minimal complications.
Adult stem cells' ease of access and lower ethical and health risks make them a more viable option for medical therapies, despite the broader differentiation potential of embryonic stem cells.
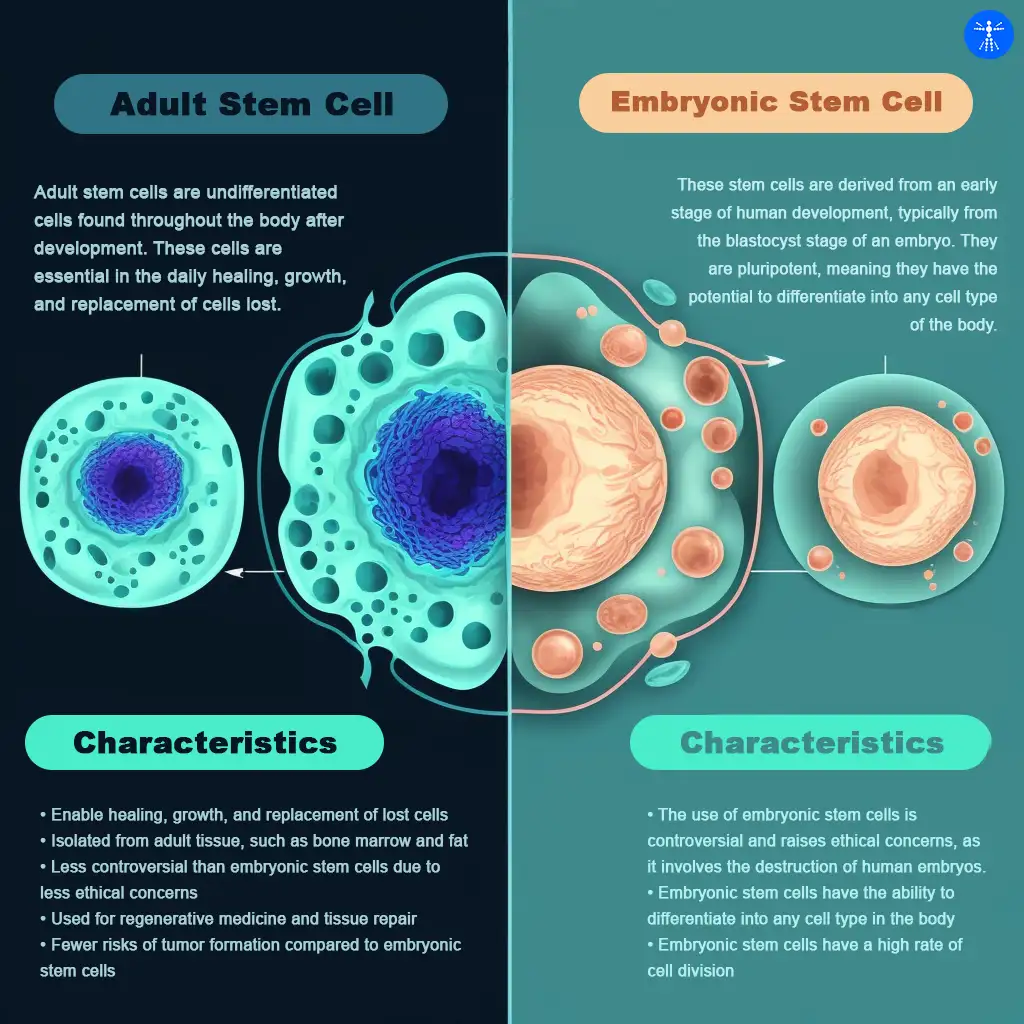
Adult stem cells (Somatic stem cells)
Somatic or adult stem cells (ASCs) are undifferentiated cells found throughout the body after development. These cells are essential in the daily healing, growth, and replacement of cells lost. Some examples include:
- Mesenchymal stem cells can become many cells in the body. MSCs can differentiate into bone, cartilage, muscle cells, brain cells, heart cells, and fat cells. They are considered pluripotent and can differentiate into cells from any germ layer.
- Neural stem cells give rise to nerve cells, brain cells, and their supporting cells, such as oligodendrocytes and astrocytes.
- Hematopoietic stem cells are blood-forming stem cells, including a red blood cell, white blood cells, and platelets. This type of stem cell is often used after bone marrow transplants. These can help people with cancer make new blood cells after their own hematopoietic progenitor cells have been killed by chemotherapy treatment. These are the only stem cells approved for use by the FDA in the US. HSCs can also be sourced from umbilical cord blood cells.
Characteristics
ASCs are undifferentiated cells found among differentiated cells in the body after development. They can self-renew and differentiate into various types of cells, depending on their location in the body. Adult pluripotent stem cells may also prove helpful in treatments; however, researchers are actively seeking methods to enhance the cultivation of these cells in laboratory settings.
They have a limited range of differentiation options compared to embryonic stem cells. Adult stem cells can be found in bone marrow, skin, and neural tissue. They play a crucial role in maintaining the normal function and repair of the body's tissues. Human stem cells can also be used for therapeutic purposes in regenerative medicine.
Research and potential use
Adult stem cells play a crucial role in the body's healing and repair processes by replenishing damaged or lost partitions. Unlike embryonic stem cells, adult cells have a more restricted range of differentiation options and can only generate specific cell types within the tissue they are found in.
Research on stem cells has led to the development of various therapeutic applications, such as using hematopoietic stem cells to treat blood cancers like leukemia and lymphoma and mesenchymal stem cells to treat bone and cartilage disorders such as osteoarthritis.
Stem cells have also been used in treating skin conditions like burns and wound healing, as well as neurological disorders like Parkinson's disease and spinal cord injury.
Clinically, adult stem cell is usually obtained from bone marrow, umbilical cord tissue, or adipose tissue through a relatively simple and minimally invasive procedure. The cells are then isolated, expanded, and differentiated in the laboratory before being reintroduced into the patient's body to treat the disease or injury.
Overall, these offer a promising approach to the development of regenerative medicine and have already shown good results in treating various diseases. However, more research is needed to fully understand the potential of a stem cell and develop safe and effective therapies for multiple conditions.
Induced pluripotent stem cells
Induced pluripotent stem cells, or iPS cells, are stem cell lines created by reprogramming a mature, specialized cell back to an embryonic stem cell-like state using specific genetic factors. The first successful generation of iPS cells in mice was created by introducing four vital genetic factors, Oct4, Sox2, Klf4, and c-Myc, into the cells using a retroviral vector.
These stem cells are produced by reprogramming adult cells, such as skin cells, to a pluripotent state. They can differentiate into any body cell type, similar to embryonic stem cells. (2)
iPS cells have been successfully derived from various cell types, including fibroblasts, pancreas, leukocytes, hepatocytes, keratinocytes, neural stem cells, cord blood cells, and more. These findings suggest that most cell types can be reprogrammed to a pluripotent state and that the average direction of cell specialization can be reversed through experimental means. Some cell types, such as neuronal progenitors, which already express one or more key reprogramming factors, may be particularly well suited for reprogramming.
Characteristics
Induced pluripotent stem (iPS) cells are stem cells generated from adult cells and reprogrammed to have characteristics similar to embryonic stem cells. These cells can differentiate into various cell types and have the potential for self-renewal. iPS cells are created by introducing specific genetic factors into adult cells, which causes them to change into a stem cell-like state.
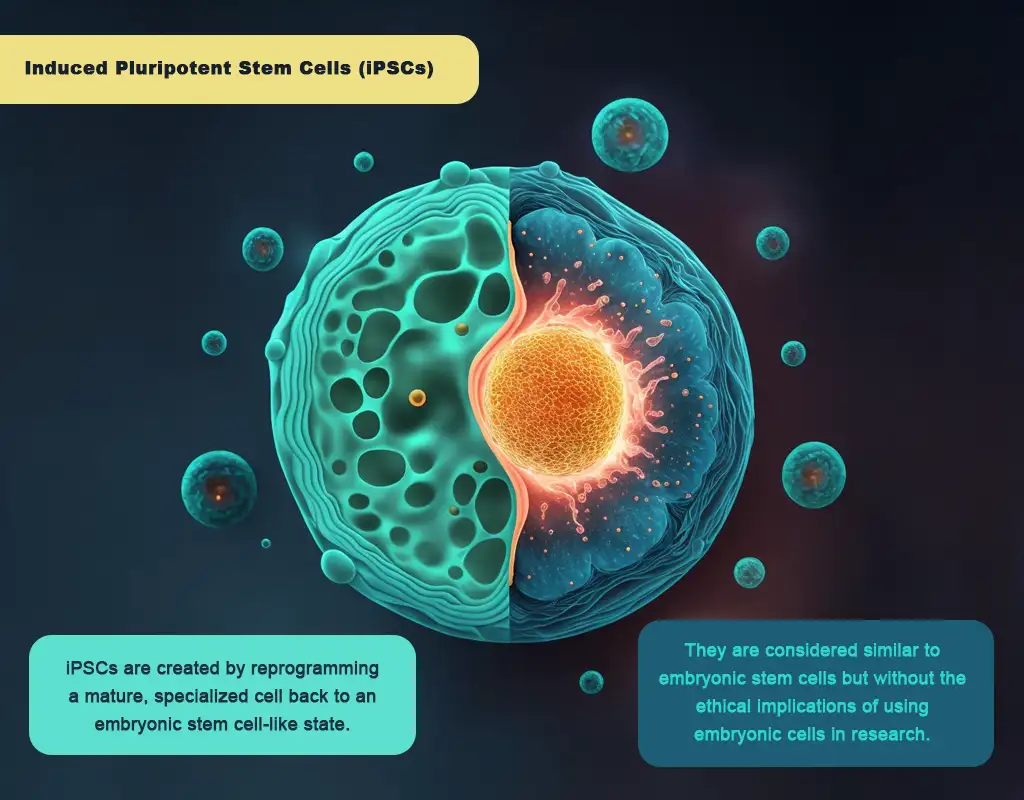
Research and potential use
These cells are considered a promising area of research due to their potential ability to be used in regenerative medicine and treat various diseases. They are considered similar to embryonic stem cells but without the ethical implications of using embryonic cells in research.
The current state of stem cell research
Studies have come a long way in the past few decades, and scientists have made significant advancements in understanding the properties and capabilities of these cells. Researchers can now isolate and culture many types of stem cells, including embryonic stem cells, somatic stem cells, and induced pluripotent stem cells.
This has allowed for a deeper understanding of the potential medical applications of stem cells and has led to the development of new treatments for various diseases and conditions.
Advancements
Recent advancements in stem cell development include the development of new techniques for isolating and culturing stem cells and creating new methods for reprogramming adult cells into a stem cell-like state.
Scientists have also made progress in understanding the factors that control stem cell differentiation, which has led to the development of new methods for controlling the differentiation of stem cells into specific cell types.
Why are stem cell lines of interest to researchers, and what purpose do they serve?
A stem cell line refers to a population of other cells derived from a single stem cell and propagated in a laboratory setting. These cells can continuously grow and remain unspecialized, meaning they do not differentiate into specific cell types, such as liver cells.
This allows for the maintenance of a healthy genetic makeup and the ability to produce more stem cells. As researchers grow stem cells, samples can be taken from the cell line for further study and research.
Potential applications
Stem cell research has the potential to revolutionize the way we treat a wide range of diseases and conditions, including cancer, heart disease, diabetes, and neurodegenerative disorders such as Alzheimer's and Parkinson's disease.
Researchers are also investigating the potential use of stem cells in regenerative medicine, which could lead to the development of new therapies for injuries and illnesses that currently have few treatment options.
Challenges and ethical considerations
Despite the many potential benefits of stem cells, many challenges must be overcome. One major challenge is the lack of understanding of the underlying biological mechanisms that govern stem cell behavior, which makes it difficult to control and predict the behavior of these cells.
Additionally, many ethical considerations are associated with stem cells, particularly when it comes to embryonic stem cells. These ethical considerations include the destruction of human embryos, the potential for creating human-animal chimeras, and the potential for abusing human clones.
Overview of stem cell therapy
Stem cell-based therapies are a treatment that uses stem cells to repair or replace damaged or diseased cells, tissues, or organs in the body. Stem cell therapies aim to regenerate damaged or lost cells and restore normal function. Stem cells are unique in their ability to differentiate into various types of cells and have the potential to repair or regenerate damaged or diseased tissue.
Types of stem cell treatments
Several stem cell treatments are being researched and developed for different medical conditions. These include:
- Hematopoietic stem cell transplantation, which uses stem cells from the bone marrow or blood to treat blood disorders such as leukemia and lymphoma
- Mesenchymal stem cell therapy, which uses stem cells from bone marrow or adipose tissue to treat conditions such as osteoarthritis, tendon injuries, and heart disease
- Embryonic stem cell therapy uses stem cells from embryos to treat a variety of conditions
- Induced pluripotent stem cell therapy, which uses adult cells that have been reprogrammed to an embryonic stem cell-like state to treat a variety of conditions
Current and potential uses
Stem cell treatment is currently used in clinical trials to treat various medical conditions, including blood disorders such as leukemia and lymphoma, bone marrow failure syndromes, and certain inherited metabolic disorders. Additionally, stem cell therapy is being researched as a potential treatment for various conditions, including heart disease, diabetes, spinal cord injury, and Alzheimer's disease.
Challenges and limitations
Despite the potential benefits of stem cell therapy, several challenges and limitations must be addressed before it can be widely adopted as a standard treatment. These include:
- Lack of standardization and regulation in the field, which can lead to inconsistent or ineffective treatments
- Difficulty in growing and maintaining more stem cells in the lab, which can limit the availability of cells for treatment
- Ethical concerns surrounding the use of embryonic stem cells can be controversial and may limit funding for research
- Safety concerns surrounding the use of stem cells, as the cells have the potential to form tumors or migrate to the wrong location in the body
- The high cost of stem cell therapy can limit access to treatment for many patients.
The importance of stem cells
Stem cells can potentially revolutionize how we treat diseases. They can differentiate into different types of cells and can be used to replace damaged or diseased cells in the body.
Somatic cell nuclear transfer and other techniques are being developed to create patient-specific stem cell lines to treat various diseases.
Future of stem cell research and therapy
With continued advancements in stem cell research, scientists are discovering new ways to use stem cells to treat diseases.
One promising area of research is using stem cells to create healthy heart muscle cells, which could be used to treat heart disease. Additionally, researchers are working on growing stem cells in the lab, making it easier to produce large numbers of cells for treatment use.
Additional resources for more information
The National Institutes of Health (NIH) has a wealth of information on stem cells, including details on the different types of stem cells, current research, and clinical trials. Additionally, the International Society for Stem Cell Research (ISSCR) is a professional organization that promotes the responsible use of stem cells in research and medicine.
For more information on using special human cells or specialized cell types to treat diseases, visit the National Marrow Donor Program (NMDP) website, which coordinates hematopoietic progenitor cell (HPC) collections and transplantation. Also, you can find many articles and journals in scientific databases such as PubMed and ScienceDirect.
References:
(1) Zakrzewski W, Dobrzyński M, Szymonowicz M, Rybak Z. Stem cells: past, present, and future. Stem Cell Res Ther. 2019 Feb 26;10(1):68. doi: 10.1186/s13287-019-1165-5. PMID: 30808416; PMCID: PMC6390367.
(2) Kumar D, Talluri TR, Anand T, Kues WA. Induced pluripotent stem cells: Mechanisms, achievements and perspectives in farm animals. World J Stem Cells. 2015 Mar 26;7(2):315-28. doi: 10.4252/wjsc.v7.i2.315. PMID: 25815117; PMCID: PMC4369489.
(3) National Institutes of Health. (2016). Stem cell basics. https://stemcells.nih.gov/info/basics.htm
(4) Mayo Clinic. (2022). Stem cells: What they are and what they do. https://www.mayoclinic.org/tests-procedures/bone-marrow-transplant/in-depth/stem-cells/art-20048117
(5) National Institutes of Health. (2016). Stem cell information. https://stemcells.nih.gov/info.htm
(6) International Society for Stem Cell Research. (n.d.). Stem cell facts. https://www.isscr.org/public-resources/stem-cell-facts
(7) Harvard Stem Cell Institute. (n.d.). About stem cells. https://hsci.harvard.edu/stem-cells-overview










